The U.S. Department of Energy has awarded $3 million to an Oregon State University College of Science researcher to lead the development of a new, high-energy-density battery that does not rely on rare elements.
Video: David Ji describes his research
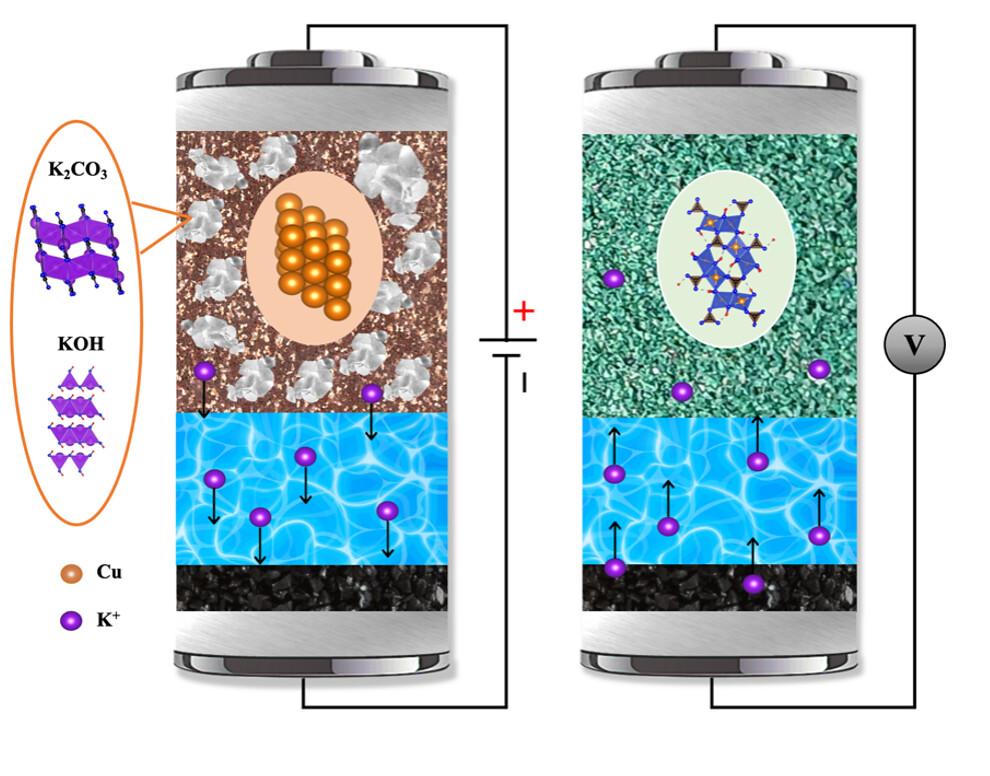
As more and more lithium-ion batteries are manufactured to electrify the transportation sector, global demand for the nickel and cobalt necessary for the batteries’ cathodes has soared, said Xiulei “David” Ji, professor of chemistry at Oregon State.
“It is predicted that there will be shortages of both elements in a matter of just decades, which means there is a pressing need to invent an alternative rechargeable battery chemistry that does not involve rare elements and potentially provides a higher energy density as well,” said Ji, who will lead a team of researchers from OSU, Howard University, the University of Maryland and Vanderbilt University.