An Oregon State University scientist and a team of undergraduate students have uncovered real-time insights into a chemical process linked with Alzheimer’s disease, paving the way toward better drug designs.
The researchers used a molecule measuring technique to observe in a laboratory setting how certain metals can promote the protein clumping that leads to the blocked neural pathways associated with Alzheimer’s.
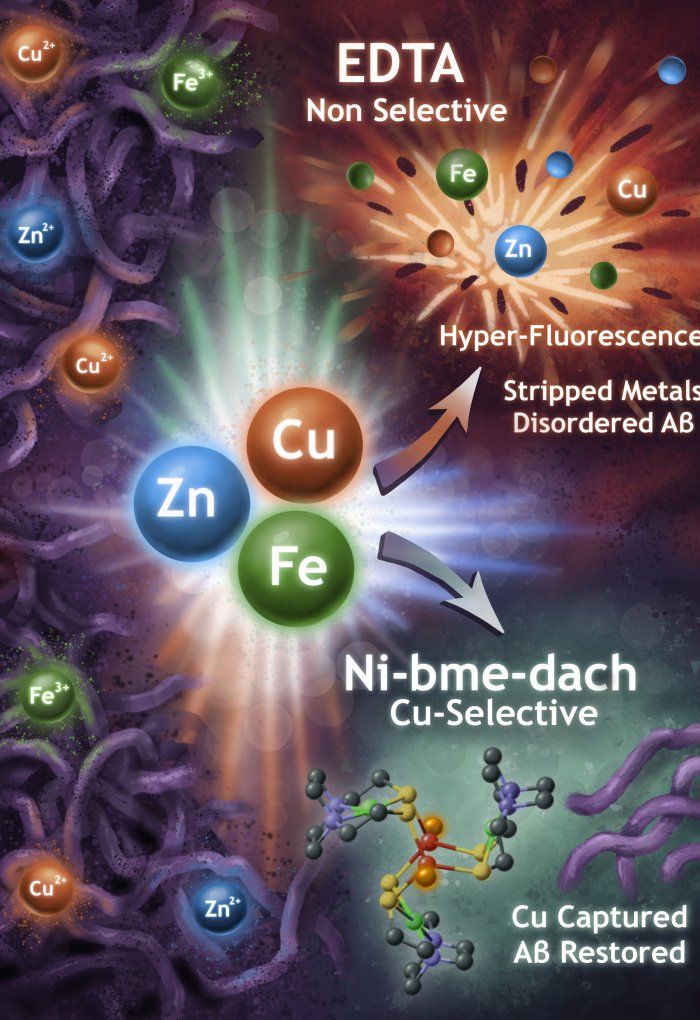
Led by Marilyn Rampersad Mackiewicz, a materials scientist and associate professor of chemistry in the OSU College of Science, the research team also watched molecules known as chelators disrupt or reverse the clumping.
Findings were published in ACS Omega.
Alzheimer’s disease is the most common form of dementia, a chronic condition of impaired cognitive function that affects large numbers of older adults and their loved ones. According to the Centers for Disease Control and Prevention, Alzheimer’s is the sixth-leading cause of death for people age 65 and older.
In Alzheimer’s patients, aggregations of amyloid-beta proteins interrupt brain cells’ ability to communicate with each other. The brain needs certain metals to work properly, but problems arise when the metals are present in unbalanced quantities.
“Too many of some metal ions, like copper, can interact with amyloid-beta proteins in ways that lead to protein aggregation, but most experiments have only shown the end result, not the interactions and aggregation process itself,” Mackiewicz said. “We developed a method that lets us observe those interactions live, second by second, and directly measure how different molecules interrupt or reverse them. It shifts the question from ‘does something work?’ to ‘how does it work, and when?’”